Current Research projects at the Winter's lab:
Vitellogenesis in the nematode Oscheius n.sp.
The nematode Oscheius n.sp. (isolate CEW1) is a free-living species isolated from the soil of São Paulo. Our interest in this nematode is centered in some aspects of its reproduction at the biochemical and molecular levels. We are currently studying its vitellogenesis. We have already characterized the proteins of the yolk from this worm (Winter, 1992). They are very similar to the proteins present in the yolk of C. elegans, being composed, at least, of three polypeptides (VT1, VT2 and VT3). Vitellins VT2 and VT3 are originated by the cleavage of a precursor coded by the gene CEW1- vit-6, and named CEW1-VIT-6. This gene is homologous to gene Ce-vit- 6 from C. elegans (Winter et al., 1996). The promotor of this gene presents the same elements already detected in the genes coding for the C. elegans vitellogenins, but not in the same number or positions. The protein coded by CEW1-vit-6 presents a higher number of amino acids substitution, in relation to homologous genes from C. elegans, that is higher at the C-terminal than at the N-terminal region of the molecule. Preliminary results obtained by Cristiane Penha-Scarabotto show that the poliadenylation site of the mRNA transcribed from CEW1-vit-6 is superposed to the termination codon. Expression of fragments from CEW1-vit-6 in E. coli have permited to determine that VT2 is at the C-terminal part of CEW1-VIT-6 (Penha, unpublished results).
Study of the SL- RNAs genes from Oscheius n.sp.
This work is being done in colaboration with the laboratory of Dr. Thomas Blumenthal currently at the University of Colorado Health Sciences Center in Denver. We have shown that Oscheius n.sp. (CEW1) present the same kind of operons present in C. elegans. Our results have shown also that the SL1-RNA genes of Oscheius n.sp. are not clustered in a single locus as has been shown in C. elegans. They are dispersed over the genome, as well as the 5S rRNA genes.
Structure and expression of an EF-1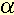 gene
gene
We have cloned a gene that codes for the subunit alpha of the elongation factor 1 of the protein synthesis. This protein has many functions in the cell, including actin polymerization and recycling of the other EF1 subunits during the elongation step of protein synthesis. This is a very conserved protein and the EF-1a from man and rabbit are 100% identical at all amino acid positions. This project is being developed by Rubens N. Akamine, a graduate student at our lab. Preliminary results have shown that unlike the EF-1a from other organisms the protein coded by this gene is not 100% conserved when compared with presumed close relatives, like C. elegans. The partial sequences obtained from this gene were used to construct a cladogram with other sequences. The resulting "tree" has a topology which is compatible with the phylogenetic relations of these organisms.
Vitellogenesis of Bothrops jararaca
We have a colaboration with the Instituto Butantan to study the proteins involved in yolk build up of B. jararaca. The Instituto Butantan has traditionally produced anti-sera against snake venoms using animals colected from nature. There is an effort on the part of this Institute to breed these snakes in captivity. Our colaboration involves the study of biochemical aspects of oocyte maturation in Bothrops jararaca. This snake is responsible for most of the snake bite accidents in Brazil. This project is being developed by Thélia R. F. Janeiro-Cinquini, a researcher at the Instituto Butantan doing her graduate studies in our lab. Our results (Janeiro-Cinquini et al., 1995) have shown, for the first time in snakes of the new world, the presence of an apolipoprotein B similar to that found in birds and mammals. Different from birds however, this protein is not taken up by the growing oocytes of B. jararaca. Unpublished results have shown that the vitellogenin of B. jararaca is composed of two dissimilar subunits that are originated from a common precursor (Janeiro-Cinquini et al., in preparation). These results will be important to determine the reproductive stage of wild and captive animals.
Last update feb/13/1997 11:11GMT